Ionic and Covalent Bonds
|
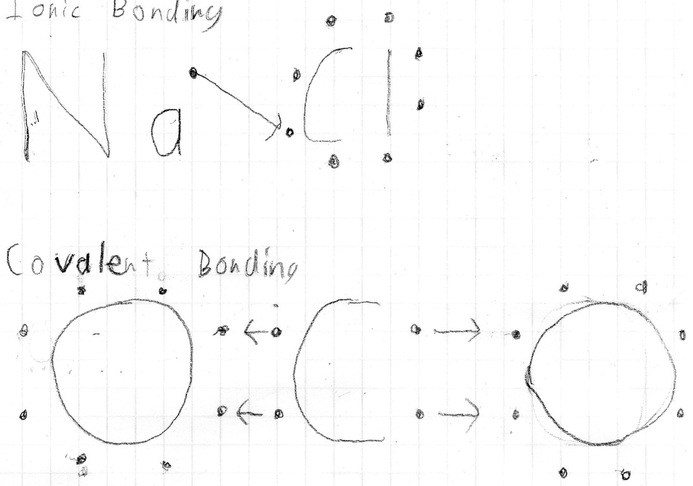
Ionic
What types of atoms form ionic bonds? Ionic bonds typically form between metals and salts. http://www.wyzant.com/resources/lessons/science/chemistry/ bonds How are ionic bonds formed and what type of structure do they create? Ionic bonds are formed when the two atoms have a large difference in terms of electronegativity, the amount of an atom’s attraction to an electron. These bonds create lattice structures. http://www.wyzant.com/resources/lessons/science/chemistry/ bonds http://hyperphysics.phy- astr.gsu.edu/hbase/chemical/bond2.html What are the typical properties of ionic substances? Include the following: physical properties, melting point, solubility in water, electrical conductivity, and any other properties you’d like to include. Ionic bonds have high melting points and high boiling points. Ionic compounds are soluble in water, and when they’re molten or have been dissolved in water, they are able to conduct electricity. The physical state of an ionic compound is solid (more specifically, crystalline solids), and they are very hard and brittle. http://hyperphysics.phy- astr.gsu.edu/hbase/chemical/bond2.html http://chemistry.about.com/od/moleculescompounds/a/Ionic- Compound-Properties.htm Short Story: Everything An Atom Always Wanted to Know About Ionic Bonding, But Was Afraid to Ask If you are reading this you're probably an atom who hasn't been able to get around to creating any lattice structures. It's OK, you needn't feel ashamed. Lot's of us come late to the party. You just haven't found the right atom yet. However, when you find your soul mate here are 4 things you should know before bonding: 1. Once you bond, you will become soluble in water, so make sure to always decline invitations to pool parties. 2. You should turn off all electricity in your house before you bond. This is the bond you create will become very conductive, and getting shocked will just put a rain on your sweet, sweet parade. 3. After you bond, both of you will begin very hard and brittle. Don't worry, it's perfectly natural and both of you will soon get used to it. 4. If you've completed your bond, now both of you will finally to get that tan without having to worry about boiling or melting. One of the many of the gifts of the ionic bond. |
Covalent
What types of atoms form covalent bonds? Covalent bonds form between two similar atoms, i.e. metal to metal. http://www.wyzant.com/resources/lessons/science/chemistry/ bonds How are covalent bonds formed and what types of structure do they create? Covalent bonds form when atoms need to get more stability. To attain this stability, the atoms combine and begin to share valence electrons. The types of bonds form molecular structures. http://chemwiki.ucdavis.edu/Theoretical_Chemistry/Chemical_ Bonding/General_Principles/Covalent_Bonds http://www2.chemistry.msu.edu/faculty/reusch/VirtTxtJml/intr o3.htm What are typical properties of covalent substances? Include the following: physical state, melting point, solubility in water, electrical conductivity, and any other properties you’d like to include. Due to covalent bond’s simple molecular structure, they are unable to conduct heat or electricity. Covalent bonds have low melting and boiling points. Covalent bonds can be gases, liquids, or solids at their physical state. These bonds are not soluble in water, but they can dissolve nonpolar liquids. http://hyperphysics.phy- astr.gsu.edu/hbase/chemical/bond2.html http://www.bbc.co.uk/schools/gcsebitesize/science/add_aqa_p re_2011/atomic/differentsubrev2.shtml Explain the difference between polar and nonpolar covalent bonds. When two atoms have a similar amount electronegativity the bond will be a nonpolar covalent bond. This is due to the fact the bond’s charge is similar on both ends. If one atom has more electronegativity than the other, then bond will be a polar covalent bond. This is because the valence electrons are more attracted to the atom with more electronegativity and will stay around that end of the bond. http://www.sciences360.com/index.php/the-difference- between-non-polar-and-polar-covalent-bonds-576/ Short Story: Everything An Atom Always Wanted to Know About Covalent Bonding, But Was Afraid to Ask If you are reading this you're probably an atom who hasn't been able to get around to creating any molecular structures. It's OK, you needn't feel ashamed. Lot's of us come late to the party. You just haven't found the right atom yet. However, when you find your soul mate here are 4 things you should know before bonding: 1. Once you bond, you will not become soluble in water, so, unlike your ionic friends, you'll actually be able to accept invites to pool parties. However, be wary of nonpolar liquids. You are soluble to those, and dissolving really puts a damper on the day. 2. You may end up as a solid, liquid, or a gas, so make sure you do your research before you bond. Always make sure you're both prepared for whatever happens next. 3. If you have more electronegativity than your bonding partner, you'll start to feel quite a few more electrons around you. This is completely normal and you should not worry about it. 4. If you've completed the bond, both of you should really stay away from the saunas. With those low melting and boiling points, nothing good will come of it. |